Put some sunshine in your engine
04 December 2020
A chemist wants to use solar energy to produce hydrogen from water. His idea? To draw inspiration from the molecules that allow plants to grow and animals to breathe.
Generating electricity or hot water from the sun is a well-known idea. However, solar energy could also be used to turn water into hydrogen or turn carbon dioxide into synthetic methane. Thus, produce “solar fuels”.

Nicolas Boscher © FNR / Rick Tonizzo
This is the approach followed by Nicolas Boscher, a researcher at the Luxembourg Institute of Science and Technology (LIST). He is developing new polymers capable of producing hydrogen in a clean and renewable way. This is crucial if it is ever to supply fuel cells to power cars, trucks and merchant ships over long distances. Nevertheless, doing so with a positive carbon balance requires being able to produce the gas cleanly, without greenhouse gas emissions.
This is difficult: hydrogen is mainly produced from methane in a reaction that emits CO2. Other processes use water, which is dissociated into hydrogen and oxygen using an electric current. Since the available electricity is partly generated from fossil fuels, this method indirectly emits carbon dioxide. As a result, hydrogen is not yet a clean fuel.
Molecules of Life
“Scientists have developed many materials to dissociate water into hydrogen from solar energy, but their efficiency is still too low or their cost too high,” notes Nicolas Boscher. “I think that a paradigm shift is needed and that entirely different approaches need to be tested”.
His inspiration comes from nature: the chemist wants to take advantage of a class of molecules crucial to life, porphyrins, which are involved in the breathing of living beings. They form the basis of haemoglobin, which transports oxygen in the blood, and chlorophyll, which enables plants to produce carbohydrates.
“These ring-shaped molecules are very versatile,” continues the researcher. “They are like Swiss Army knives for a chemist like me.”
The first stage of Nicolas Boscher’s project consists of creating porphyrin polymers and weaving them into a three-dimensional network. This extremely porous material allows water molecules to pass through. These molecules can then interact with the porphyrins. By absorbing sunlight, their ring produces an electron that dissociates water into its two components, hydrogen and oxygen.

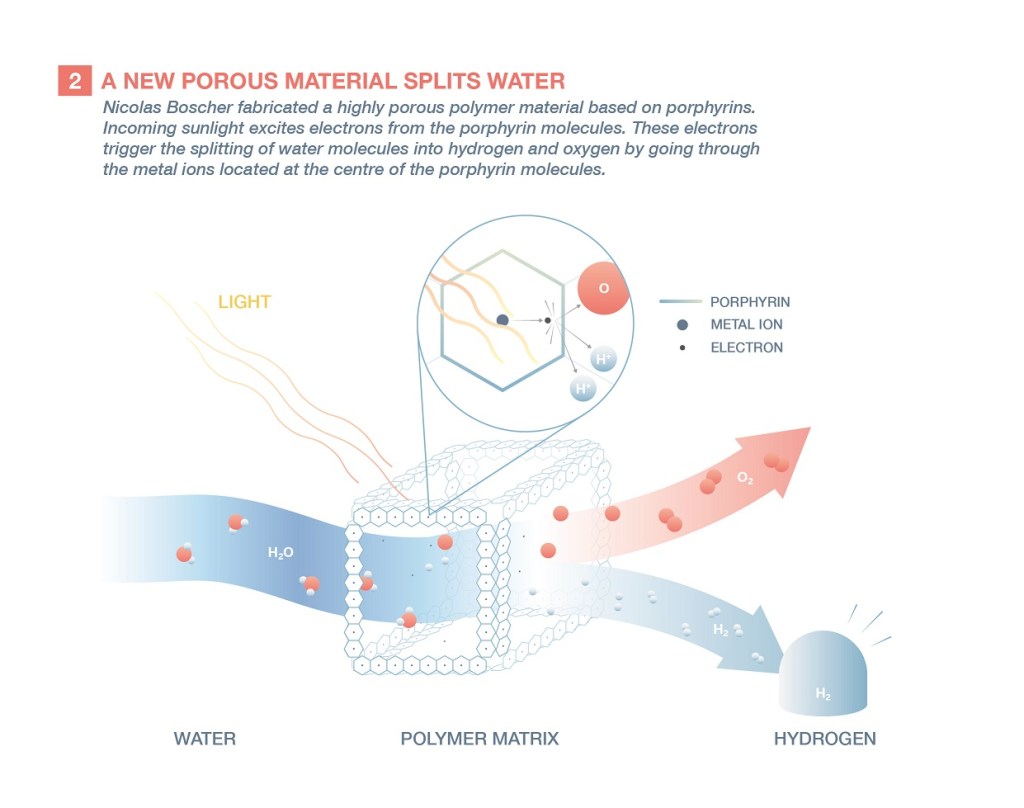
Chemical synthesis in a vacuum
If the idea seems simple, implementing it is less so. Because porphyrins, insoluble in water, are difficult to handle by traditional chemistry based on reactions in liquids. The scientist uses a different type of process: he reacts the different components by mixing them in gaseous form at very low pressure.
“This technique, chemical vapour deposition, is typically used in the manufacture of electronic chips. I did my first research in this field, which allowed me to adapt these methods to work with organic components, including porphyrins.”
The team has already been able to make thin porous layers that join two hydrogen atoms into a gas molecule. It still must be combined with the preliminary step of dissociating the water into hydrogen atoms and gaseous oxygen.
Towards a greener chemistry
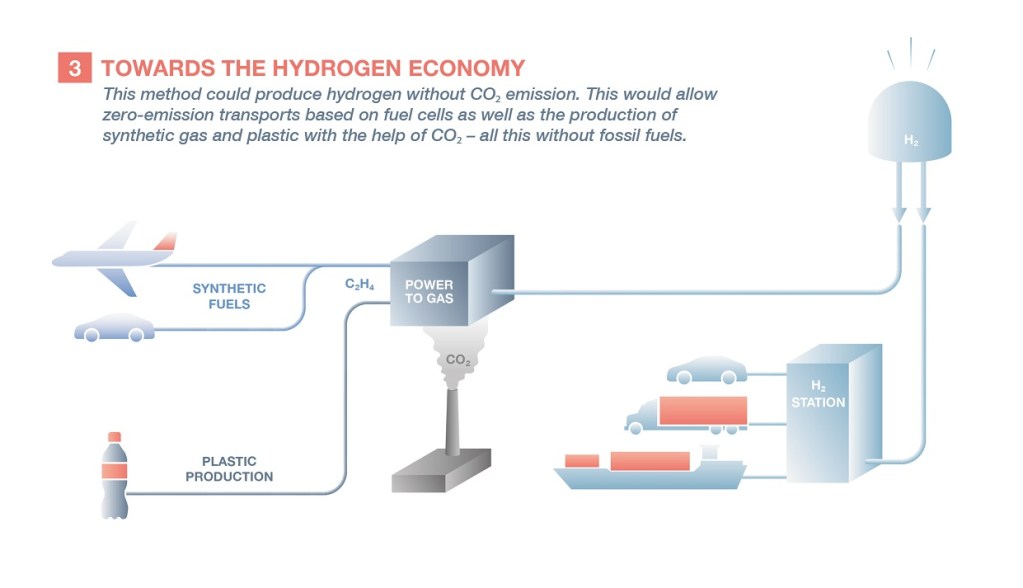
The renewable production of hydrogen can also be used to produce synthetic methane and plastics, which generally come from the fossil fuel sector. This requires combining it with carbon atoms, obtained by extracting CO2 from the ambient air or by filtering the gases emitted in thermal power stations. This would represent a giant step towards green chemistry, which does not use fossil fuels either as a source of energy or as a raw material.
This article was originally published by the Luxembourg National Research Fund
About the European Research Council (ERC)
The European Research Council, set up by the EU in 2007, is the premiere European funding organisation for excellent frontier research. Every year, it selects and funds the very best, creative researchers of any nationality and age, to run projects based in Europe. The ERC offers four core grant schemes: Starting, Consolidator, Advanced and Synergy Grants. With its additional Proof of Concept grant scheme, the ERC helps grantees to bridge the gap between grantees’ pioneering research and early phases of its commercialisation. https://erc.europa.eu/









